
Julien C, Anakok E, Treton X, Nachury M, Nancey S, Buisson A, Fumery M, Filippi J, Maggiori L, Panis Y, Zerbib P, François Y, Dubois A , Sabbagh C, Rahili A, Seksik P, Allez M , Lefevre JH , REMIND Study Group Investigators, Le Corff S, Bonnet A , Beyer-Berjot L , Sokol H
J Crohns Colitis 2022 Feb 26;jjac026.
doi: 10.1093/ecco-jcc/jjac026.
Background: Surgery is performed in 50-70% of Crohn’s disease (CD) patients, and its main risk is surgical site infection (SSI). The microbiota has been extensively assessed in CD but not as a potential risk factor for septic morbidity. The objective of this study was to assess the impact of the gut microbiota on SSI in CD. Methods: We used the multicentric REMIND prospective cohort to identify all patients who experienced SSI after ileocolonic resection for CD, defined as any postoperative local septic complication within 90 days after surgery: wound abscess, intra-abdominal collection, anastomotic leakage, enterocutaneous fistula. The mucosa-associated microbiota of the ileal resection specimen was analyzed by 16S sequencing in 149 patients. The variable selection and prediction were performed with random forests (R package VSURF) on clinical and microbiotal data. The criterion of performance that we considered was the area under the Receiver Operating Characteristic (ROC) curve (AUC). Results: SSI occurred in 24 patients (16.1%), including 15 patients (10.1%) with major morbidity. There were no significant differences between patients with or without SSI regarding alpha and beta diversity. The top selected variables for the prediction of SSI were all microbiota-related. The maximum AUC (0.796) was obtained with a model including 14 genera, but an AUC of 0.78 was already obtained with a model including only six genera (Hungatella, Epulopiscium, Fusobacterium, Ruminococcaceae_ucg_009, Actinomyces, and Ralstonia). Conclusion: The gut microbiota has the potential to predict SSI after ileocolonic resection for CD. It might play a role in this frequent postoperative complication.
Figure : Bacterial taxa decreased or increased in surgical site infection
Microbiota in neuroinflammation and synaptic dysfunction: a focus on Alzheimer’s disease
Bairamian D, Sha S, Rolhion N, Sokol H, Dorothée G, Lemere CA, Krantic S
Mol Neurodegener. 2022 Mar 5;17(1):19.
DOI: 10.1186/s13024-022-00522-2
Abstract Background: The implication of gut microbiota in the control of brain functions in health and disease is a novel, currently emerging concept. Accumulating data suggest that the gut microbiota exert its action at least in part by modulating neuroinflammation. Given the link between neuroinflammatory changes and neuronal activity, it is plausible that gut microbiota may affect neuronal functions indirectly by impacting microglia, a key player in neuroinflammation. Indeed, increasing evidence suggests that interplay between microglia and synaptic dysfunction may involve microbiota, among other factors. In addition to these indirect microglia-dependent actions of microbiota on neuronal activity, it has been recently recognized that microbiota could also affect neuronal activity directly by stimulation of the vagus nerve. Main messages: The putative mechanisms of the indirect and direct impact of microbiota on neuronal activity are discussed by focusing on Alzheimer’s disease, one of the most studied neurodegenerative disorders and the prime cause of dementia worldwide. More specifically, the mechanisms of microbiota-mediated microglial alterations are discussed in the context of the peripheral and central inflammation cross-talk. Next, we highlight the role of microbiota in the regulation of humoral mediators of peripheral immunity and their impact on vagus nerve stimulation. Finally, we address whether and how microbiota perturbations could affect synaptic neurotransmission and downstream cognitive dysfunction. Conclusions: There is strong increasing evidence supporting a role for the gut microbiome in the pathogenesis of Alzheimer’s disease, including effects on synaptic dysfunction and neuroinflammation, which contribute to cognitive decline. Putative early intervention strategies based on microbiota modulation appear therapeutically promising for Alzheimer’s disease but still require further investigation.
Figure : Hypothetical link between gut dysbiosis and mechanisms leading to the pathogenesis of AD.

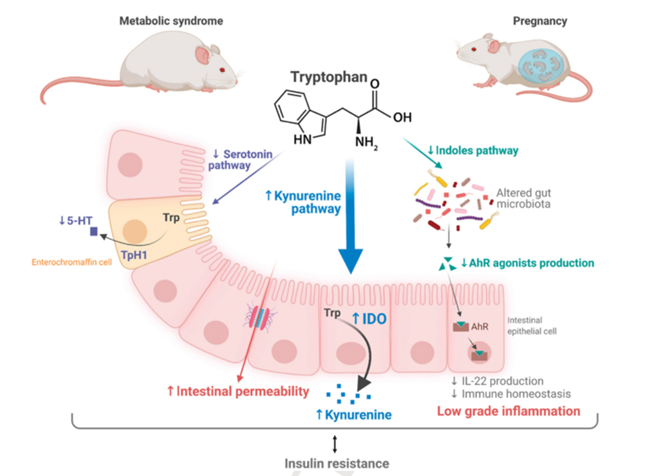
Gut Microbiota Reprogramming f Tryptophan Metabolism During Pregnancy Shapes Host Insulin Resistancepect
Gastroenterology. 2022 Mar 2; S0016-5085(22)00215-3.

Cholesterol-to-Coprostanol Conversion by the Gut Microbiota: What We Know, Suspect, and Ignore
Catherine Juste, Philippe Gérard
Every day, up to 1 g of cholesterol, composed of the unabsorbed dietary cholesterol, the biliary cholesterol secretion, and cholesterol of cells sloughed from the intestinal epithelium, enters the colon. All cholesterol arriving in the large intestine can be metabolized by the colonic bacteria. Cholesterol is mainly converted into coprostanol, a non-absorbable sterol that is excreted in the feces. Interestingly, cholesterol-to-coprostanol conversion in human populations is variable, with a majority of high converters and a minority of low or inefficient converters. Two major pathways have been proposed, one involving the direct stereospecific reduction of the Δ5 double bond direct while the indirect pathway involves the intermediate formation of 4-cholelesten-3-one and coprostanone. Despite the fact that intestinal cholesterol conversion was discovered more than a century ago, only a few cholesterol-to-coprostanol-converting bacterial strains have been isolated and characterized. Moreover, the responsible genes were mainly unknown until recently. Interestingly, cholesterol-to-coprostanol conversion is highly regulated by the diet. Finally, this gut bacterial metabolism has been linked to health and disease, and recent evidence suggests it could contribute to lower blood cholesterol and cardiovascular risks.

Cholesterol-to-Coprostanol Conversion by the Gut Microbiota: What We Know, Suspect, and Ignore
Catherine Juste, Philippe Gérard
Every day, up to 1 g of cholesterol, composed of the unabsorbed dietary cholesterol, the biliary cholesterol secretion, and cholesterol of cells sloughed from the intestinal epithelium, enters the colon. All cholesterol arriving in the large intestine can be metabolized by the colonic bacteria. Cholesterol is mainly converted into coprostanol, a non-absorbable sterol that is excreted in the feces. Interestingly, cholesterol-to-coprostanol conversion in human populations is variable, with a majority of high converters and a minority of low or inefficient converters. Two major pathways have been proposed, one involving the direct stereospecific reduction of the Δ5 double bond direct while the indirect pathway involves the intermediate formation of 4-cholelesten-3-one and coprostanone. Despite the fact that intestinal cholesterol conversion was discovered more than a century ago, only a few cholesterol-to-coprostanol-converting bacterial strains have been isolated and characterized. Moreover, the responsible genes were mainly unknown until recently. Interestingly, cholesterol-to-coprostanol conversion is highly regulated by the diet. Finally, this gut bacterial metabolism has been linked to health and disease, and recent evidence suggests it could contribute to lower blood cholesterol and cardiovascular risks.

Interactions between diet and gut microbiota are critical regulators of energy metabolism. The efects of fbre intake have been deeply studied but little is known about the impact of proteins. Here, we investigated the efects of high protein supplementation (Investigational Product, IP) in a double blind, randomised placebo-controled intervention study (NCT01755104) where 107 participants received the IP or an isocaloric normoproteic comparator (CP) alongside a mild caloric restriction. Gut microbiota profles were explored in a patient subset (n= 53) using shotgun metagenomic sequencing. Visceral fat decreased in both groups (IP group:− 20.8 ± 23.2 cm2 ; CP group: − 14.5 ± 24.3 cm2 ) with a greater reduction (p< 0.05) with the IP supplementation in the Per Protocol population. Microbial diversity increased in individuals with a baseline low gene count (p< 0.05). The decrease in weight, fat mass and visceral fat mass signifcantly correlated with the increase in microbial diversity (p< 0.05). Protein supplementation had little efects on bacteria composition but major diferences were seen at functional level. Protein supplementation stimulated bacterial amino acid metabolism (90% aminoacid synthesis functions enriched with IP versus 13% in CP group (p< 0.01)). Protein supplementation alongside a mild energy restriction induces visceral fat mass loss and an activation of gut microbiota amino-acid metabolism.

Butyrate, a new microbiota-dependent player in CD8+ T cells immunity and cancer therapy?
The intestinal microbiota is a new promising avenue in cancer immunotherapy, but mechanisms remain elusive. He et al. demonstrate that butyrate, a bacterial metabolite, enhances the CD8+ T cell response and improve chemotherapy efficacy through ID2-dependent IL-12 signaling.

Gut interleukin-17A (IL-17)-producing γδ T cells are tissue-resident cells that are involved in both host defense and regulation of intestinal inflammation. However, factors that regulate their functions are poorly understood. In this study, we find that the gut microbiota represses IL-17 production by cecal γδ T cells. Treatment with vancomycin, a Gram-positive bacterium-targeting antibiotic, leads to decreased production of short-chain fatty acids (SCFAs) by the gut microbiota. Our data reveal that these microbiota-derived metabolites, particularly propionate, reduce IL-17 and IL-22 production by intestinal γδ T cells. Propionate acts directly on γδ T cells to inhibit their production of IL-17 in a histone deacetylase-dependent manner. Moreover, the production of IL-17 by human IL-17-producing γδ T cells from patients with inflammatory bowel disease (IBD) is regulated by propionate. These data contribute to a better understanding of the mechanisms regulating gut γδ T cell functions and offer therapeutic perspectives of these cells.

Gut microbiota-derived metabolites as central regulators in metabolic disorders
Metabolic disorders represent a growing worldwide health challenge due to their dramatically increasing prevalence. The gut microbiota is a crucial actor that can interact with the host by the production of a diverse reservoir of metabolites, from exogenous dietary substrates or endogenous host compounds. Metabolic disorders are associated with alterations in the composition and function of the gut microbiota. Specific classes of microbiota-derived metabolites, notably bile acids, short-chain fatty acids, branched-chain amino acids, trimethylamine N-oxide, tryptophan and indole derivatives, have been implicated in the pathogenesis of metabolic disorders. This review aims to define the key classes of microbiota-derived metabolites that are altered in metabolic diseases and their role in pathogenesis. They represent potential biomarkers for early diagnosis and prognosis as well as promising targets for the development of novel therapeutic tools for metabolic disorders.


Recipient factors in faecal microbiota transplantation: one stool does not fit all
Faecal microbiota transplantation (FMT) is a promising therapy for chronic diseases associated with gut microbiota alterations. FMT cures 90% of recurrent Clostridioides difficile infections. However, in complex diseases, such as inflammatory bowel disease, irritable bowel syndrome and metabolic syndrome, its efficacy remains variable. It is accepted that donor selection and sample administration are key determinants of FMT success, yet little is known about the recipient factors that affect it. In this Perspective, we discuss the effects of recipient parameters, such as genetics, immunity, microbiota and lifestyle, on donor microbiota engraftment and clinical efficacy. Emerging evidence supports the possibility that controlling inflammation in the recipient intestine might facilitate engraftment by reducing host immune system pressure on the newly transferred microbiota. Deciphering FMT engraftment rules and developing novel therapeutic strategies are priorities to alleviate the burden of chronic diseases associated with an altered gut microbiota such as inflammatory bowel disease.

Paracellular permeability of the intestinal epithelium is a feature of the intestinal barrier, which plays an important role in the physiology of gut and the whole organism. Intestinal paracellular permeability is controlled by complex processes and is involved in the passage of ions and fluids (called pore pathway) and macromolecules (called leak pathway) through tight junctions, which seal the intercellular space. Impairment of intestinal paracellular permeability is associated with several diseases. The identification of a defect in intestinal paracellular permeability may help to understand the implication of gut barrier as a cause or a consequence in human pathology. Here we describe two complementary methods to evaluate alteration of paracellular permeability in cell culture, using the human intestinal cell line Caco-2 and its clone Caco-2/TC7.

The current pandemic of coronavirus disease (COVID) 2019 constitutes a global public health issue. Regarding the emerging importance of the gut-lung axis in viral respiratory infections, analysis of the gut microbiota’s composition and functional activity during a severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection might be instrumental in understanding and controling COVID 19. We used a nonhuman primate model (the macaque), that recapitulates mild COVID-19 symptoms, to analyze the effects of a SARS-CoV-2 infection on dynamic changes of the gut microbiota. 16S rRNA gene profiling and analysis of β diversity indicated significant changes in the composition of the gut microbiota with a peak at 10-13 days post-infection (dpi). Analysis of bacterial abundance correlation networks confirmed disruption of the bacterial community at 10-13 dpi. Some alterations in microbiota persisted after the resolution of the infection until day 26. Some changes in the relative bacterial taxon abundance associated with infectious parameters. Interestingly, the relative abundance of Acinetobacter (Proteobacteria) and some genera of the Ruminococcaceae family (Firmicutes) was positively correlated with the presence of SARS-CoV-2 in the upper respiratory tract. […]

Use of Ussing Chambers to Measure Paracellular Permeability to Macromolecules in Mouse Intestine
An increased intestinal permeability has been described in many diseases including inflammatory bowel disease and metabolic disorders, and a better understanding of the contribution of intestinal barrier impairment to pathogenesis is needed. In recent years, attention has been paid to the leak pathway, which is the route of paracellular transport allowing the diffusion of macromolecules through the tight junctions of the intestinal epithelial lining. While the passage of macromolecules by this pathway is very restricted under physiological conditions, its amplification is thought to promote an excessive immune activation in the intestinal mucosa. The Ussing chambers have been widely used to measure both active and passive transepithelial fluxes in intact tissues. In this chapter we present how this simple device can be used to measure paracellular permeability to macromolecules in the mouse intestine. We propose a detailed protocol and describe how to best exploit all the possibilities of this technique, correctly interpret the results, and avoid the main pitfalls.

Tryptophan metabolites get the gut moving
Nicolas Benech, Nathalie Rolhion, Harry Sokol
How gut microbes can regulate the enteric nervous system and gut-brain communications is a field of intense research. In this issue of Cell Host & Microbe, Ye et al. demonstrate that bacteria can control intestinal motility and vagal neuronal activation via tryptophan catabolites through the receptor TrpA1 of enteroendocrine cells.

Changes in the intestinal microbiome have been associated with obesity and type 2 diabetes, in epidemiological studies and studies of the effects of fecal transfer in germ-free mice. We review the mechanisms by which alterations in the intestinal microbiome contribute to development of metabolic diseases, and recent advances, such as the effects of the microbiome on lipid metabolism. Strategies have been developed to modify the intestinal microbiome and reverse metabolic alterations, which might be used as therapies. We discuss approaches that have shown effects in mouse models of obesity and metabolic disorders, and how these might be translated to humans to improve metabolic health.

Tryptophan Metabolism as a Pharmacological Target
L-Tryptophan is an essential amino acid required for protein synthesis. It undergoes an extensive and complex metabolism along several pathways, resulting in many bioactive molecules acting in various organs through different action mechanisms. Enzymes involved in its metabolism, metabolites themselves, or their receptors, represent potential therapeutic targets, which are the subject of dynamic research. Disruptions in L-tryptophan metabolism are reported in several neurological, metabolic, psychiatric, and intestinal disorders, paving the way to develop drugs to target it. This review will briefly describe L-tryptophan metabolism and present and discuss the most recent pharmacological developments targeting it.

Non-alcoholic fatty liver diseases (NAFLD) are associated with changes in the composition and metabolic activities of the gut microbiota. However, the causal role played by the gut microbiota in individual susceptibility to NAFLD and particularly at its early stage is still unclear. In this context, we transplanted the microbiota from a patient with fatty liver (NAFL) and from a healthy individual to two groups of mice. We first showed that the microbiota composition in recipient mice resembled the microbiota composition of their respective human donor. Following administration of a high-fructose, high-fat diet, mice that received the human NAFL microbiota (NAFLR) gained more weight and had a higher liver triglycerides level and higher plasma LDL cholesterol than mice that received the human healthy microbiota (HR). Metabolomic analyses revealed that it was associated with lower and higher plasma levels of glycine and 3-Indolepropionic acid in NAFLR mice, respectively. Moreover, several bacterial genera and OTUs were identified as differently represented in the NAFLR and HR microbiota and therefore potentially responsible for the different phenotypes observed. Altogether, our results confirm that the gut bacteria play a role in obesity and steatosis development and that targeting the gut microbiota may be a preventive or therapeutic strategy in NAFLD management.

Gut microbiota-derived metabolites as central regulators in metabolic disorders
Metabolic disorders represent a growing worldwide health challenge due to their dramatically increasing prevalence. The gut microbiota is a crucial actor that can interact with the host by the production of a diverse reservoir of metabolites, from exogenous dietary substrates or endogenous host compounds. Metabolic disorders are associated with alterations in the composition and function of the gut microbiota. Specific classes of microbiota-derived metabolites, notably bile acids, short-chain fatty acids, branched-chain amino acids, trimethylamine N-oxide, tryptophan and indole derivatives, have been implicated in the pathogenesis of metabolic disorders. This review aims to define the key classes of microbiota-derived metabolites that are altered in metabolic diseases and their role in pathogenesis. They represent potential biomarkers for early diagnosis and prognosis as well as promising targets for the development of novel therapeutic tools for metabolic disorders.

The Gut Microbiota at the Service of Immunometabolism
The gut microbiota is implicated in immune system functions. Regulation of the metabolic processes occurring in immune cells is crucial for the maintenance of homeostasis and immunopathogenesis. Emerging data demonstrate that the gut microbiota is an actor in immunometabolism, notably through the effect of metabolites such as short-chain fatty acids, bile acids, and tryptophan metabolites. In this Perspective, we discuss the impact of the gut microbiota on the intracellular metabolism of the different subtypes of immune cells, including intestinal epithelial cells. Besides the effects on health, we discuss the potential consequences in infection context and inflammatory bowel diseases.

Declaration of conflicting interests: The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: H.S. received unrestricted study grants from Danone, Biocodex and Enterome, and board membership, consultancy or lecture fees from Carenity, Abbvie, Astellas, Danone, Ferring, Mayoly Spindler, MSD, Novartis, Roche, Tillots, Enterome, Maat, BiomX, Biose, Novartis and Takeda. He is also a co-founder of Exeliom Biosciences. N.B. received a travel grant from Maat.
